Feraheme’s Label Expansion is an Opportunity to Impact LOS Outcomes for Hospital Inpatients with IDA
In Feraheme, Iron Deficiency Anemia (IDA)
Get Dexur’s Personalized Hospital Specific Presentation on Quality, Safety, Compliance & Education
By: Jeffrey Maser Feb. 26, 2018
The FDA’s recent approval of label expansion for Feraheme (ferumoxytol injection), by AMAG Pharmaceuticals, could be an opportunity for hospitals to positively impact patient length of stay outcomes. Feraheme, which was previously approved only for Chronic Kidney Disease (CKD) has now been expanded to all patients with Iron Deficiency Anemia (IDA). The company’s press release following label expansion states a few key points:
- The Feraheme label expansion approval was supported by two positive pivotal Phase 3 trials evaluating Feraheme versus iron sucrose or placebo in a broad population of patients with IDA. It was also supported by positive results from a third Phase 3 randomized, double-blind, clinical safety trial comparing Feraheme to Injectafer® (ferric carboxymaltose injection) in approximately 2,000 adults with IDA. The study demonstrated comparability to Injectafer® based on the primary composite endpoint of the incidence of moderate-to-severe hypersensitivity reactions (including anaphylaxis) and moderate-to-severe hypotension.
- Nik Grund, chief commercial officer at AMAG said: “An expanded Feraheme label that includes all eligible adult IDA patients will double the addressable market opportunity estimated to be nearly a million patients per year for Feraheme and provide for future growth within this broader IDA market segment, In addition, roughly 4.5 million Americans suffer from the debilitating effects of IDA. AMAG is proud that Feraheme may now be a treatment option for all eligible IDA patients.”
What are the implications of the Feraheme label expansion regarding quality outcomes in hospitals?
Researchers, hospital quality and pharmacy teams have been studying the effects of IDA on quality outcomes for a number of years and have found an increase in length of stay (LOS) among other impacted outcomes. A 2010 Stanford literature review of 49 papers on Anemia and Blood Management in Hips and Knees found four studies that showed an increased LOS for patients with anemia. Specifically, the literature found the following:
- Anemia on admission and postoperative anemia were both associated with a significantly increased LOS of the same order of magnitude in all four prospective cohort studies reporting this clinical outcome. In anemic patients compared with nonanemic patients, mean LOS was 18 versus 11 days (P 0.001), 13 versus 8 days (P 0.001), and 16 versus 8 days (P 0.01). Finally, Gruson et al. reported without quantifying their findings that anemia at hospital admission was significantly associated with an increased hospital LOS (P 0.01).
Dexur’s Hospital and IDN specific IDA Quality Outcomes database, based on Medicare claims data, also shows similar LOS differences for patients who develop IDA post-admission in the hospital.
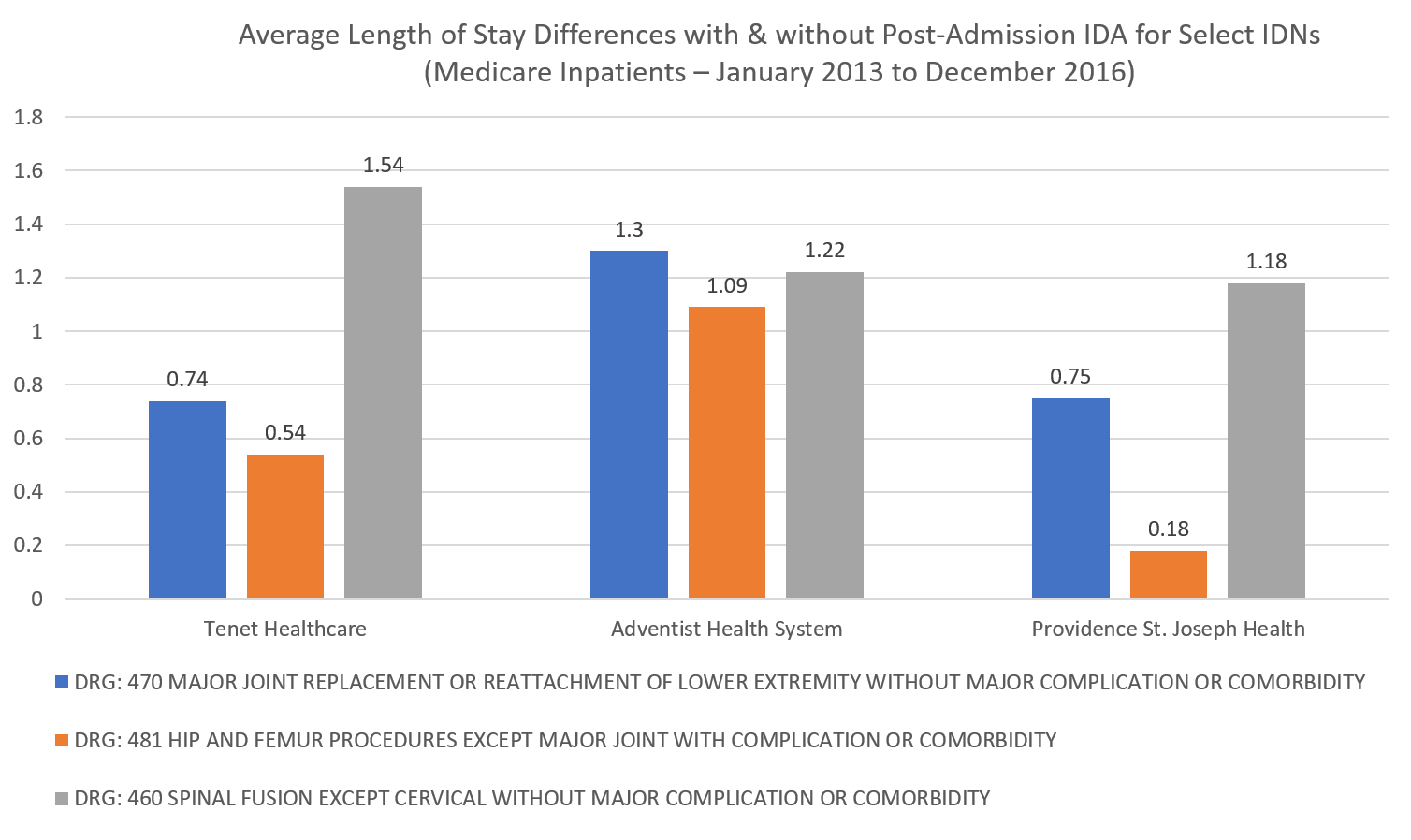
DRG-470, DRG-481 and DRG-460 all showed a higher average LOS when IDA was developed post-admission within the hospital stay. For example, DRG-481 (Hip and Femur Procedures with CC) shows that three different systems (Tenet, Adventist & Providence St. Joseph) had a difference in LOS ranging from 0.18 to 1.09 days. This wide variation in LOS metrics for IDA patients show that there is still significant opportunity for IV Iron solutions like Feraheme and Injectafer to positively improve hospital quality outcomes.
Further References:
ABOUT THE AUTHOR
