Melinta’s Baxdela could Make an Impact on Hospital ABSSSI Readmissions: An Analysis of Florida Hospital Orlando Data
In Baxdela, Acute Bacterial Skin and Skin Structure Infections (ABSSSI)
Get Dexur’s Personalized Hospital Specific Presentation on Quality, Safety, Compliance & Education
By: Jeffrey Maser Mar. 19, 2018
Baxdela, by Melinta Therapeutics, was recently approved by the FDA and has been launched to treat serious bacterial infections. Baxdela (delafloxacin) was approved for the treatment of adult patients with acute bacterial skin and skin structure infections (ABSSSI) caused by gram-positive and gram-negative organisms. A common example of a gram-positive organism is Methicillin-resistant Staphylococcus aureus (MRSA) while an example of a widely known gram-negative organism is Escherichia coli (E.coli). The options for treating ABSSSI have been limited but Baxdela was found to be effective in treating ABSSSI compared to the standard combined treatment of antibiotics vancomycin and aztreonam.
The clinical trials in regards to efficacy were similar between Baxdela and the vancomycin/aztreonam arm, where positive responses to the treatments were defined as a decrease in lesion size of 20% or greater. However, safety results favored Baxdela compared to the administration of vancomycin and aztreonam. At least one treatment-emergent adverse event was found to occur 60.4% of the time for the vancomycin/aztreonam arm compared to only a 49.5% rate for Baxdela. Eugene Sun, MD, CEO of Melinta, echoed this finding by explaining to Infectious Disease News that “There is an advantage to being able to accomplish the same thing with one drug vs. two drugs. You are better off from the side effect standpoint.”
The effective treatment of skin infections presents a challenging problem across the healthcare continuum and has substantial impact on quality as well as the economic burden within hospitals. Dexur analysts highlighted Medicare inpatient skin infection discharges at Florida Hospital Orlando, one of the highest volume hospitals for skin infections in the nation. Of the hospital’s 156,046 total discharges between January 2013 - December 2016, 10,667 discharges (6.84%) were associated with a skin infection. Diving a bit deeper, Dexur’s data reveals that among the total discharges, 1.96% of patients receiving care at the hospital were readmitted within 30 days for a skin infection, on par with the national average of 1.91%.
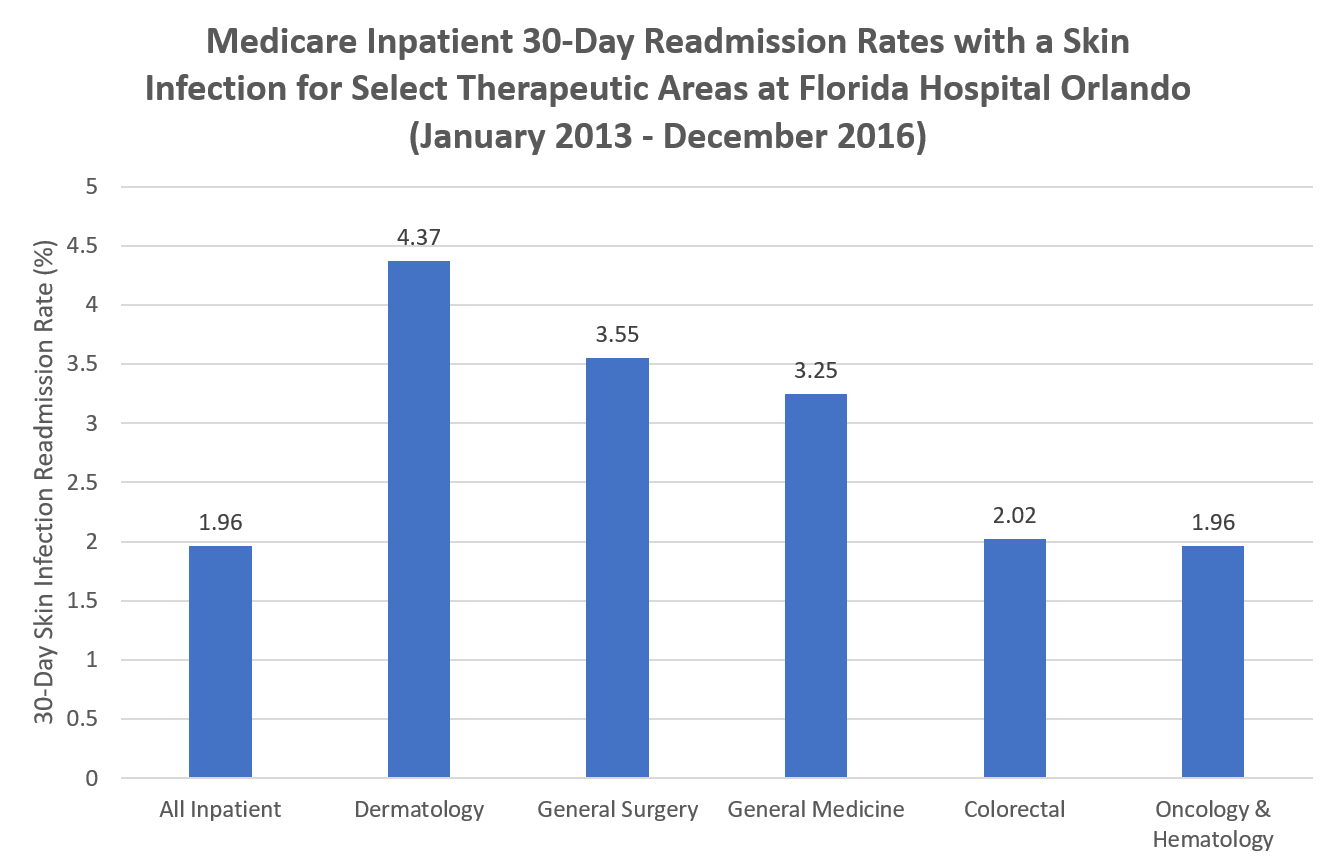
Readmissions with a skin infection were most common for dermatology discharges, as readmissions occurred for 6.4% of dermatology discharges from Florida Hospital Orlando within 30 days. Specific diagnosis related groups within this discipline were associated with even higher readmission rates. One example is DRG-602 (Cellulitis With Major Complication or Comorbidity), where the readmission rate with skin infections at Florida Hospital Orlando was 12.04% between 2013-2016. However, readmissions with skin infections were not only common among dermatology discharges. For instance, at the state level, skin infection readmissions were found to occur for 3.97% of the DRG-158 (Dental And Oral Diseases With Complication or Comorbidity) discharges for all Florida Medicare enrollees. Another example is DRG-945 (Rehabilitation With Complication or Comorbidity/Major Complication or Comorbidity), where 3.31% of discharges at the state level had a skin infection readmission. Such readmissions can have a significant impact on hospital quality and economic outcomes so effective treatment of skin infections would be a great boon to hospitals and physicians across the U.S. 30-day readmission rates for discharges with skin infection readmissions can be found for multiple therapeutic areas in the below table.
DEXUR PRO MEMBERS GET ACCESS TO:
- Florida Hospital Orlando Discharge Volume, Florida Hospital Orlando 30-Day Skin Infection Readmissions, Florida Hospital Orlando 30-Day Skin Infection Readmission Rate, Florida 30-Day Skin Infection Readmission Rate and National 30-Day Skin Infection Readmission Rate by Therapeutic Area between January 2013 - December 2016
ABOUT THE AUTHOR
