Baxdela’s Potential Impact on Readmissions for Patients with ABSSSI: An Analysis of Beaumont Hospital, Royal Oak Data
In Baxdela, Acute Bacterial Skin and Skin Structure Infections (ABSSSI)
Get Dexur’s Personalized Hospital Specific Presentation on Quality, Safety, Compliance & Education
By: James Pitt Apr. 19, 2018
Baxdela (delafloxacin), by Melinta Pharmaceuticals, is a fluoroquinolone antibiotic recently approved for treatment of acute bacterial skin and skin structure infections (ABSSSI). Dexur previously examined Baxdela’s potential for readmissions in Florida. Clinical trials in March 2018 and December 2017 found lower rates of treatment discontinuation with Baxdela than with vancomycin plus aztreonam. This is significant because initial treatment failure is common in serious skin infections. According to a 2011 retrospective study, initial antibiotic therapy failed in 22.4% of patients with ABSSSI.
The FDA defines ABSSSI as a bacteria skin infection with a lesion size of at least 75 cm2, which is approximately 3.5 by 3.5 inches. Previously, similar infections were designated cSSSIs (complicated skin and skin structure infections). A 2013 study in Surgical Infections on cSSSIs found that initial treatment failed in 16.6% of acute infections, 34.1% of chronic/ ulcerative infections, and 26.7% of surgical site infections.
Dexur examined readmissions associated with skin infections at Beaumont Hospital in Royal Oak, Michigan, one of the highest volume hospitals for dermatology in the nation. Of the hospital’s 91,543 Medicare inpatient discharges between January 2013 and December 2016, there were 22,217 30-day readmissions. 1,783 of these related to skin infections.
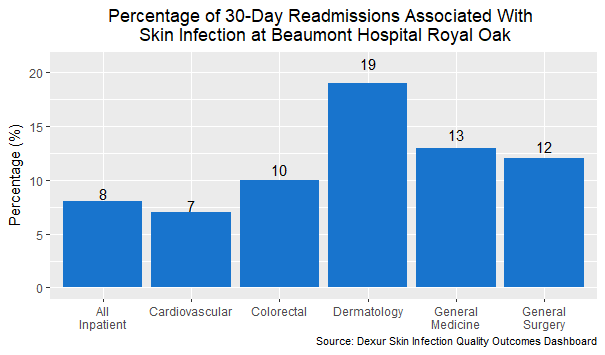
The dermatology department had the highest percentage of skin infection readmissions, no surprise as dermatological cases already concern the skin. A closer analysis of general surgery readmissions reveals something more surprising. 12% of general surgery readmissions involved skin infections. This rose to 16% for DRGs 867-868 (other infectious and parasitic diseases) and 18% for DRGs 862-863 (postoperative and post-traumatic infections).
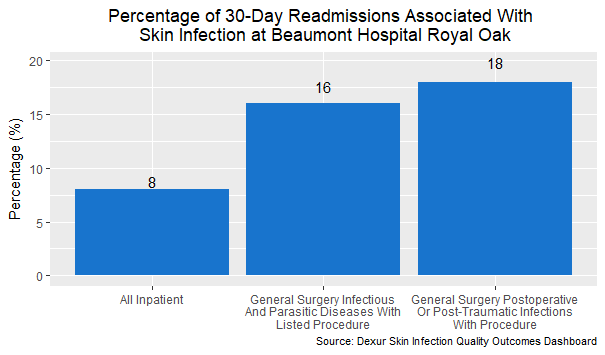
This suggests that recurring infections after treatment may pose a particular challenge. An antibiotic with lower treatment discontinuation, such as Baxdela, could help reduce these readmissions.
DEXUR PRO MEMBERS GET ACCESS TO:
- Beaumont Hospital, Royal Oak Total Discharges, All-Cause Readmissions, and Skin Infection Readmissions for all departments 2013-2016.
- Hospital, state, and national comparisons of skin infection readmission rate as a percentage of all discharges.
ABOUT THE AUTHOR
